How antiviral from Hepatitis C could damage other viruses
By Andy Fell
A new virus-killing peptide springs from an unexpected source: another virus, Hepatitis C.
Now biomedical engineers at UC Davis and Nanyang Technological University, Singapore show how the HCV alpha-helical (AH) peptide can make holes in the types of membranes that surround viruses. The work is published Jan. 5 in Biophysical Journal.
HCV-AH is known to be active against a wide range of viruses including West Nile, dengue, measles and HIV.
The HCV-AH peptide appears to target an Achilles’ heel common to many viruses, most likely a property of the lipid coating or envelope, said study author Atul Parikh, professor of biomedical engineering at UC Davis. That means that it’s less likely that viruses can readily evolve to become resistant to the peptide.
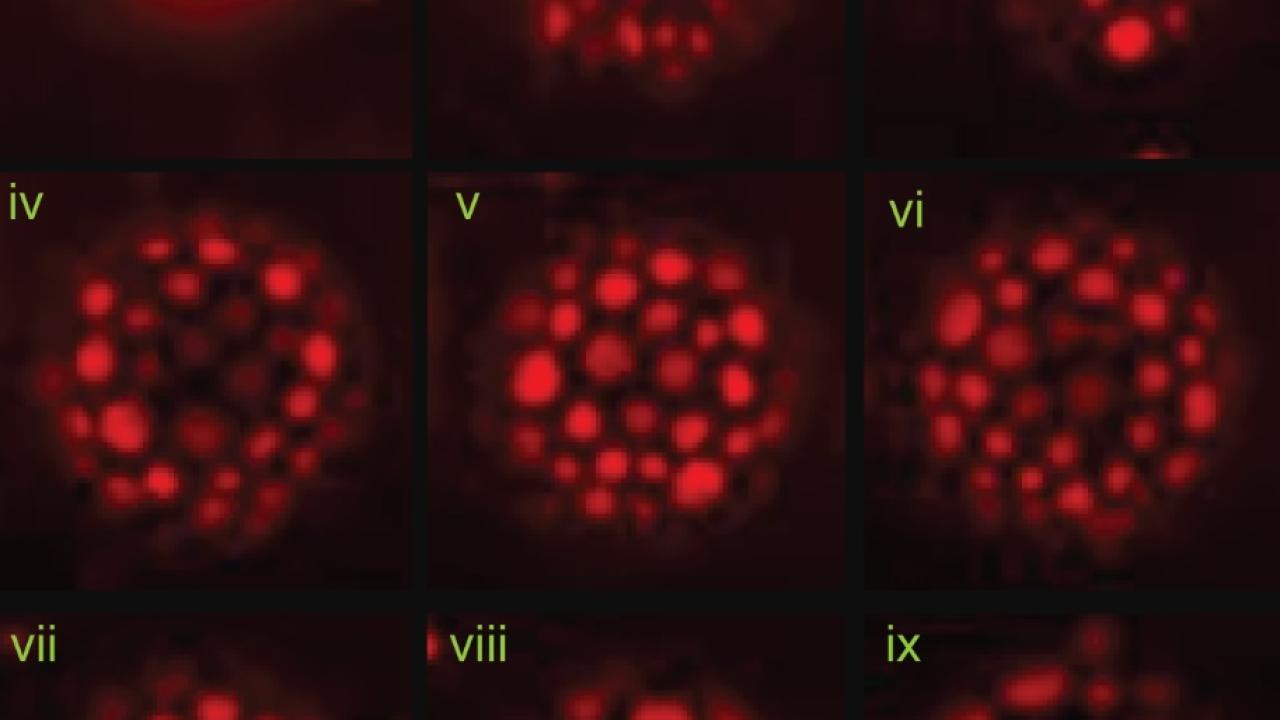
Parikh, Nam-Joon Cho of Nanyang Technological University and colleagues tested the properties of HCV-AH with simplified model lipid membranes. Essentially, these are tiny “soap bubbles” made up of a layer of lipids, just like living cells and viruses, but without the cellular contents.
The HCV-AH peptide had different effects depending on the composition of the membrane. When the membranes were rich in cholesterol, like those of many viruses, the peptide caused the membrane lipids to clump together forming bright spots under the microscope. But cholesterol-free membranes did not show the same effect.
Video: “Bubbling” shows membranes failing as a result of exposure to AH peptide
Additional experiments showed that the peptide also had different effects depending on the size of the vesicle.
There are currently no antiviral drugs that work by destabilizing the virus membrane, Cho said, although some have been proposed.
The researchers now plan to move to study the effects of the peptide on more complex membranes and then live human cells and viruses. If the mechanism still seems promising, it could eventually move into preclinical testing.
“Understanding how the drug candidate interacts with these biologically important lipids, we reason, should open the door to deciphering the rich and complex biology of these systems and lead to new opportunities for antiviral strategies,” Parikh said.
The work was supported by the U.S. Department of Energy, the National Research Foundation and the National Medical Research Council of Singapore, and Nanyang Technological University.
Original post here: http://blogs.ucdavis.edu/egghead/2016/01/05/how-antiviral-from-hepatitis-c-could-damage-other-viruses/
