UC Davis Researchers Strengthen Stem-Cell Transplants for Better Patient Outcomes
New cell-combination strategy creates longer transplant viability
UC Davis researchers have discovered a promising new way to use a combination of two types of stem cells to improve transplantation viability – and possible patient outcomes.
As outlined in Stem Cell Research & Therapy, the UC Davis team took on a long-standing challenge for stem-cell researchers: stem cells’ potentially short transplant lives.
“Cells can easily get killed after transplantation if they don’t have any special capability to deal with the environment at the transplantation site,” explained senior author of the article Dr. Aijun Wang of the UC Davis Biomedical Engineering Department and School of Medicine (Surgery Department). “By co-transplanting two types of stem cells, we found that we can achieve long-term engraftment.”
The team used hemophilia A as a model for their study’s efforts to strengthen stem-cell transplants. The disease is a genetic disorder caused by a single missing or defective blood protein. Affecting approximately 1 in every 5,000 male births, this potentially dangerous genetic disorder causes blood to not clot properly, leading to spontaneous bleeding as well as bleeding following injuries or surgery. This single-gene disorder had been considered a good model for focused cell-based gene therapy, but efforts to address it through the use of potentially therapeutic cells had been hampered by the cells’ quick demise upon transplantation.
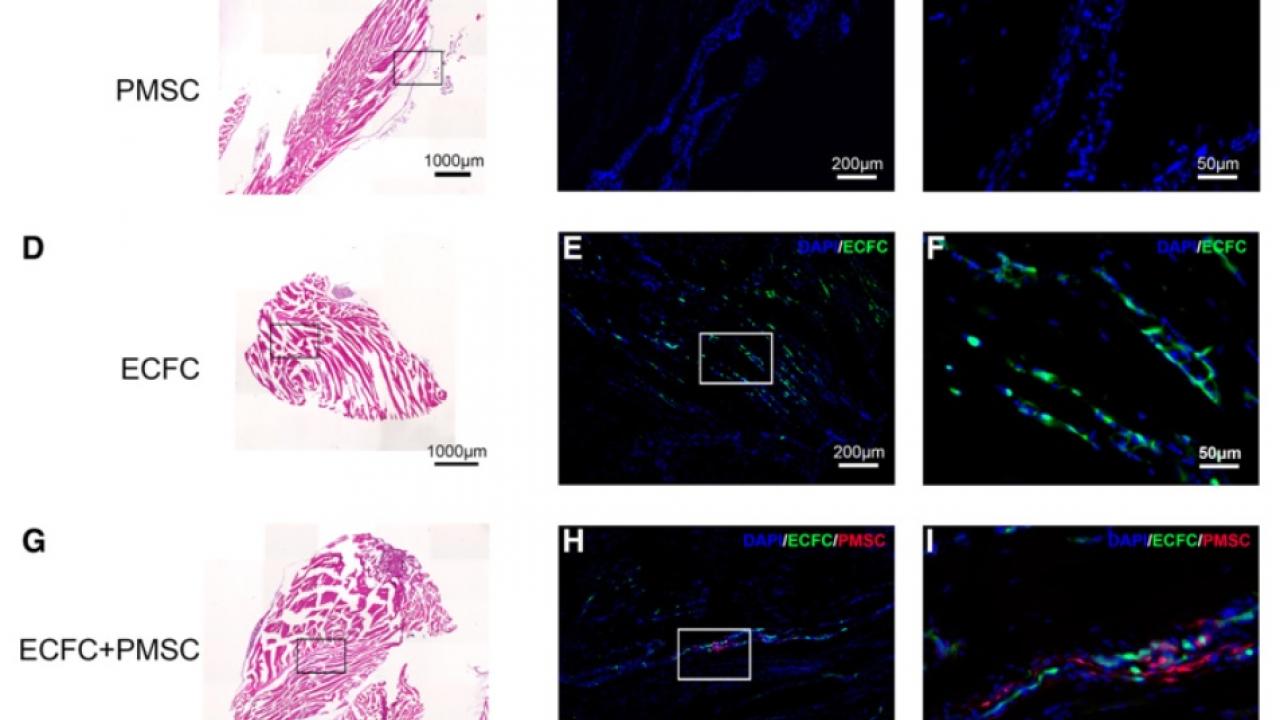
But by using two types of stem cells -- endothelial colony-forming cells (ECFCs) and placenta-derived mesenchymal stromal cells (PMSCs) – the UC Davis team found they could achieve the type of long-term engraftment that had long eluded researchers.
“By using this particular combination of stem cells to newborn mice, we were able to lengthen the survival of transplanted stem cells to over six months, which is a long time considering the typical mouse lifespan,” said Wang. “Researchers have always been trying to achieve longer engraftment after stem-cell transplantation – it’s been a long-standing problem. With this specifically designed cotransplantation, we can achieve long-term engraftment using hemophilia as a model where the cells are not only engrafted but also are functional in correcting the disease phenotype.”
Team members plan to continue related research to see how this key finding could be applied in human treatments for hemophilia and other genetic disorders.
“Our overall goal is to treat patients with genetic or structural birth defects as early as possible and, with our ongoing stem-cell work on hemophilia, spina bifida and other disorders and birth defects, we’re moving in that direction,” said Wang. “Hopefully we can reach the point where we can address and cure these birth defects even before the baby is born – we’re working really hard to make these goals a reality!”
